What are the particular features of phospholipids? In a nutshell: they have multifunctional effects, they are physiological, produced naturally in the body, biodegradable and they are free of adverse effects. Beyond that, they are components of our daily nutrition or contained in food supplements. In spite of their relatively high molecular mass, they are readily absorbed by the skin. Their history dates back to the beginnings of life on our planet.
Biomembranes
With the evolution of biomembranes from phospholipids, the first microorganisms could organize in cells and seal themselves off from the outside world. As the communication with the outside world and the transport of nutrients had to be ensured, the cellular biomembranes had developed a selective permeability that has been continually optimized in the course of time with the help of proteins integrated for this purpose. This particular set-up has not been changed until today. Hence phospholipids are ubiquitous where there are living cells, not only in microorganisms but also in all herbal and animal forms of life. Phospholipid extraction is a rather complicated process and the base materials used today are egg yolk and soya lecithin (majority). Soya lecithin more or less is a waste product in the soya oil production.
Compositions
Phospholipids consist of a glycerin, fatty acids and phosphoric acid containing backbone which is chemically bound to variable amino- or poly alcohols as e.g. choline, ethanolamine, glycerin, inositol, and serine.1 The most important phospholipids occurring in mammalian membranes are:2
- phosphatidylcholine (PC; membrane fraction: 45-55%)
- phosphatidylethanolamine (PE; membrane fraction: 15 -25%)
- phosphatidylinositol (PI; membrane fraction: 10-15%)
- phosphatidylserine (PS; membrane fraction: 5-10%)
- phosphatidic acid (PA; membrane fraction: 1-2%); PA has no alcohol component
Further membrane components are cholesterol (CH; 10-20%), sphingomyelin (SM; 5-10%) and cardiolipin (CL; 2-5%), which is a phosphatidylglycerin (PG)-derived component of the intracellular mitochondria membranes.
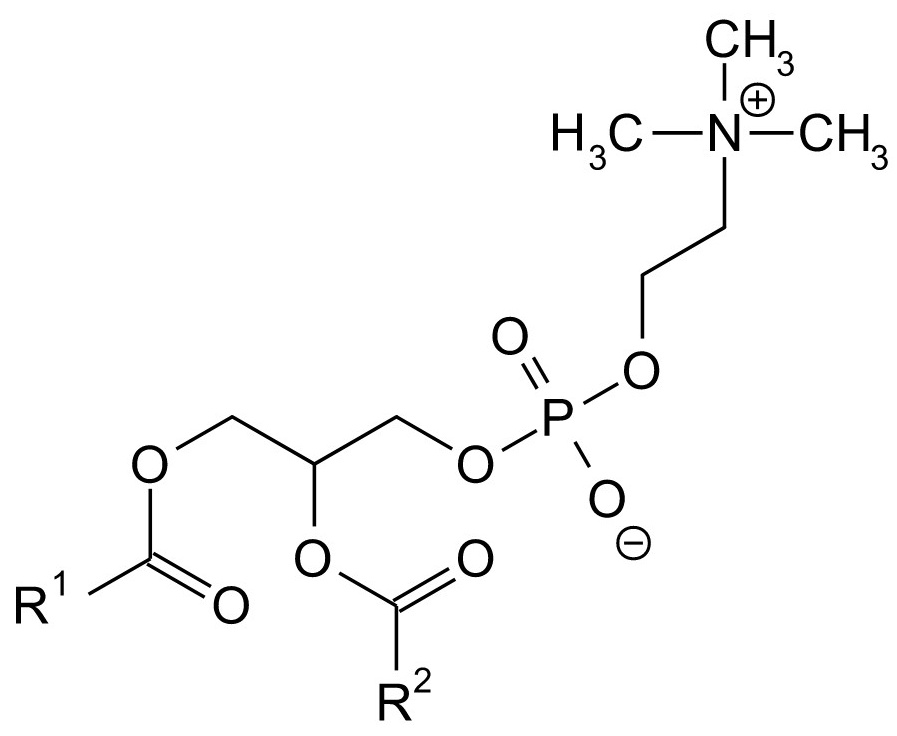
Phosphatidylcholine
Phosphatidylcholine (PC) gained from soya lecithin is most frequently used in topical preparations. Lecithin is a natural, weak anionic emulsifier used in food industry for the preparation of mayonnaise, bakery improvers and chocolates, among others. By contrast, PC spontaneously forms spherical membranes (vesicles) which are in accordance with the former herbal cells in terms of size and shape.
Carriers - liposomes
The vesicles can encapsulate water-soluble substances in their interior but also integrate lipophilic substances in the membranes. The resulting dispersions, discovered for the first time in the sixties3, do not require emulsifiers. Different versions have later on been used for the liposomes technology4,5. In contrast to the initial thinking that liposomes cannot only penetrate into the skin but also permeate through the skin, it could subsequently be proved that liposome membranes only fuse with the intercellular membrane structure of the stratum corneum and thus improve the penetration. In this process the phase transition temperature of the skin barrier or in other words, the transition from the less permeable coherent lamellar gel phase6 into a more permeable liquid-crystalline phase is lowered. The phase transition temperature of soya PC is below 0°C due to the high content of essential fatty acids.
Lamellar base creams
By hydrogenating essential fatty acids into saturated fatty acids, this particular property of soya PC is lost. Hydrogenated phosphatidylcholine, albeit in combination with sterines (cholesterol, phytosterines) and ceramides can, figuratively speaking, be used as a filler for the interrupted gel phase membranes of the horny layer7 in the case of dry skin since the phase transition temperature of about 42°C is close to the transition temperatures of the stratum corneum8. In technical terms, we speak of lamellar base creams with derma membrane structure. They are used for skin protection and skin care purposes and are characterized by an extremely low washout effect compared to conventional emulsions. The term washout-effect means the loss of cream- and skin barrier components during body cleansing - already when using plain water.
Nanodispersions
Since liposomes have a limited absorption capacity for lipophilic substances, biodegradable nanodispersions have been developed with particles of similar size which are surrounded by single instead of double membranes. Phosphatidylcholine nanoparticles are liquid and suited for the encapsulation and application of essential fatty acids (linoleic acid, alpha- and gamma-linolenic acid) in the form of linseed-, kiwi- and evening primrose oil, as well as lipophilic vitamins and their derivatives. Nanodispersions also improve the penetration of encapsulated active agents.
Troubleshooters
The carriers (liposomes, nanodispersions) consisting of PC themselves are a very effective active agent and suited for the treatment of acne9 (15-lipoxygenase-substrate10) and of barrier disorders (ceramide I-substrate11) due to their high fraction of bound linoleic acid and the low content of alpha-linolenic acid. Since the PC of the epidermis and the sphingomyelines (SM) - participating in the composition of membranes, as mentioned above - are in balance from which then ceramides develop during the apoptosis of skin cells, the skin will regenerate and improve after a topical application of external PC.12.13 The following overview shows typical combinations of PC with active agents. The indications listed refer to therapy (dermatological) and supportive prevention (cosmetic) respectively the adjuvant skin care.
| PC carrier |
Active agent |
Indication |
Mode of action |
| liposomes |
ascorbyl phosphate (vitamin C) |
anti-aging, hyperpigmentation |
collagen synthesis↑, tyrosinase inhibition, radical scavenger14,15 |
| liposomes |
azelaic acid (according to the German Federal Institute for Risk Assessment (BfR)16 up to 1%) |
acne, rosacea, perioral dermatitis |
5-alpha-reductase-inhibitor (bacteria)17 |
| nanodispersion |
boswellic acids |
acne, rosacea, inflammation |
protease inhibition18 |
| nanodispersion |
coenzyme Q10 |
anti-aging |
radical-scavenger15, lipid metabolism↑ |
| liposomes |
caffeine |
cellulite |
lipolysis19, microcirculation↑ |
| liposomes |
Euphrasia Officinalis extract |
eye care
|
eyebright (also angel's eye, bird's eye) - popular medicine |
| liposomes |
fumaric acid |
psoriasis |
drug fumitory (also earth smoke, common fumitory) - popular medicine |
| liposomes |
hyaluronic acid (combination, not encapsulated) |
anti-aging |
skin hydration↑, wrinkle reduction |
| liposomes |
isoflavones |
anti-aging |
local estrogenic effects20 |
| liposomes |
Kigelia africana fruit extract |
anti-aging |
skin tightening |
| nanodispersion |
kiwi oil |
burns, sun burn, inflammation |
15-lipoxygenase-substrate21 |
| liposomes |
Laminaria digitata (wakame, also oar kelp) |
anti-aging |
skin hydration↑, wrinkle reduction |
| nanodispersion |
linseed oil |
burns, sun burn, inflammation |
15-lipoxygenase-substrate21 |
| nanodispersion |
evening primrose oil |
neurodermitis, inflammation |
15-lipoxygenase-substrate, substitute in the case of a delta-6-desaturase-defect21 |
| liposomes |
NMF (amino acids) |
anti-aging |
skin hydration↑, radical scavenger22 |
| liposomes |
proanthocyanidin (OPC) |
anti-aging |
radical-scavenger15 |
| nanodispersion |
retinol acetate or retinyl palmitate (vitamin A) |
anti-aging, acne,
|
regeneration14, collagen synthesis↑ |
| liposomes |
sphingosin-1-phosphate |
psoriasis |
inhibition of keratinocyte proliferation23 |
| liposomes |
spilanthol |
anti-aging |
wrinkle reduction through muscle relaxation24 |
| nanodispersion |
tocopherolacetate (vitamin E) |
skin protection, anti-aging |
radical-scavenger15, regeneration14 |
| liposomes |
tranexamic acid |
hyperpigmentation, rosacea |
tyrosinase inhibition, anti-fibrinolysis25 |
liposomes
|
niacinamide (vitamin B3) |
anti-aging |
regeneration14, anti-inflammatory |
liposomes
|
zinc salts |
acne |
component of oxidoreductases21 |
The choline fraction (2-hydroxyethyl-trimethylammonium salt) of PC and SM is the base substance for acetylcholine, a neurotransmitter, which is essential for brain and nerve functions.26,27 In addition, choline is a significant methyl group donor in the human metabolism.
Phosphatidylcholine-based oleogels
PC is easy to integrate into the lamellar barrier layers of the skin, a property which is also used in the context of mineral oil free oleogels. The products are non-aqueous and have a content of more than 90% of herbal lipids and fatty acids. Despite their high lipid content, the oleogels have an excellent penetrability and can be used for the care of atopic skin, for infantile skin care and, when formulated with appropriate active agents, also for the care of rosacea skin. Since there is no water phase which has to be stabilized with cosmetic additives such as emulsifiers and preservatives, tolerance problems as for instance allergies or irritations are virtually excluded. Further medical indications are perioral dermatitis, perianal barrier disorders, diabetic skin and decubitus (bet sores).28
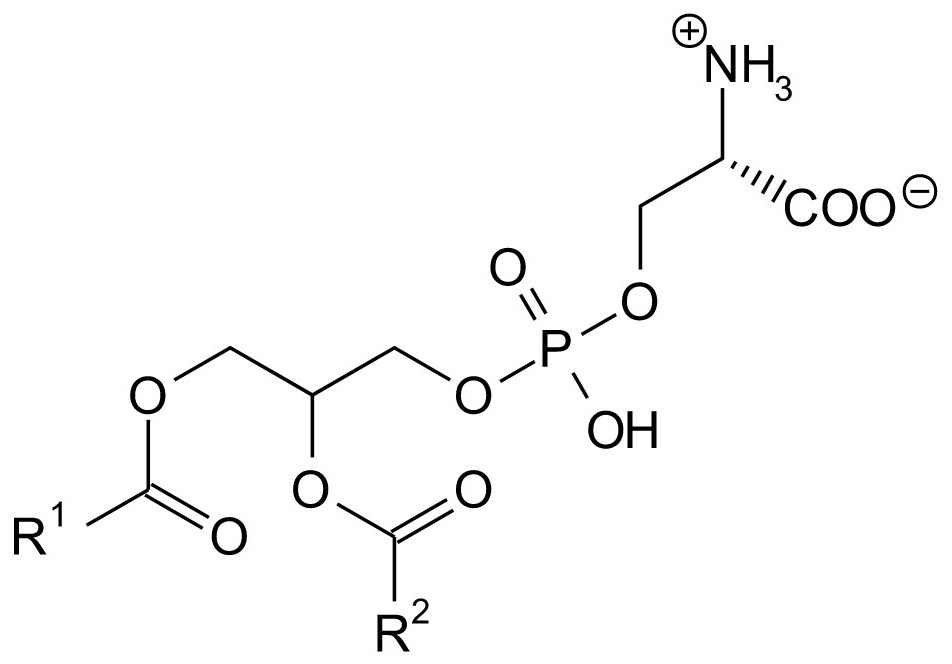
Phosphatidylserine
Phosphatidylserine (PS) belongs to the vital phospholipids29 and is found in the interior part of the bilayered cell membrane of herbal and animal cells. With apoptosis or in other words, the programmed cell death, PS enzymatically finds its way to the outward part of the membrane. This is a signal for macrophages (the scavenger cells or phagozytes of the immune system) to encircle and digest the respective cells30. With injuries and the thus related blood coagulation, phosphatidylserine also ends up at the cell surface and accelerates the blood coagulation.31 Since it activates the macrophages and the blood coagulation, phosphatidylserine has an anti-inflammatory effect.32,33 In this context, also neuroprotective34, antioxidative32 effects, and the inhibition of UV-induced procollagen degradation and MMP-135 formation are discussed.36
When hydrogenated phosphatidylcholine is substituted by unsaturated anionic soya phosphatidylserine (PS), lamellar creams are converted into water-in-oil emulsions (W/O). Since phosphatidylserine is a physiological substance and perfectly fits into the natural phospholipid balance of the body, these emulsions do not cause washout effects. The contrary is the case: the emulsions have an excellent adhesiveness and the transepidermal water loss (TEWL) can be reduced with the processed lipid substances. The creams have anti-inflammatory effects due to phosphatidylserine as messenger but also due to the chemically bound essential fatty acids in phosphatidylserine. Similar to the unsaturated soya phosphatidylcholine, the latter mentioned are released in the skin through the phospholipases A1 and A2 and subsequently converted into anti-inflammatory metabolites through 15-lipoxygenase (15-LOX).37,10 Phosphatidylserine creams can be used for skin protection purposes, in the case of eczema, light sun erythema and for the care of the atopic skin. Interesting fact: higher concentrations of phosphatidylserine occur in cold water fish (herring, mackerel) and in the cerebral matter.38 PS-enriched extracts are sold as food supplements. With calcium salts, phosphatidylserine forms tubular structures (cochleates) that, in nanoscale, are suitable carriers for pharmaceutical drugs.
Synthetic phospholipids
The properties of natural phospholipids are the basis for a multitude of synthetic variants used as liposome base substances. Their purpose is a modified degradation of the carriers and the controlled availability of pharmaceutical drugs in different routes of administration (topical, intravenous, oral).
Figures
Structural formula of phosphatidylcholine
Structural formula of phosphatidylserine
References
- Lautenschläger H, Phospholipide - Multitalente, medical Beauty Forum 2014;3:18-20
- van Hoogevest P, Phospholipids - Properties, manufacturing and use, 5th International Symposium on "Phospholipids in Pharmaceutical Research", Heidelberg 2017
- Bangham AD, Horne RW, Negative staining of phospholipids and their structural modification by surface-active agents as observed in the electron microscope, Journal of Molecular Biology 1964;8;5:660-668
- Sessa G, Weissmann G, Phospholipid spherules (liposomes) as a model for biological membranes, Journal of Lipid Research 1968;9;3:310-318
- Lautenschläger H, Handbook of Cosmetic Science and Technology edited by Barel AO, Paye M and Maibach HI, CRC Press Taylor & Francis Group, Boca Raton 2006:155-163
- Norlén L, Skin barrier structure and function: the single gel phase model, J Invest Dermatol. 2001 Oct;117(4):830-6
- Iwai I et al, The human skin barrier is organized as stacked bilayers of fully extended ceramides with cholesterol molecules associated with the ceramide sphingoid moiety, J Invest Dermatol 2012 Sep;132(9):2215-25
- Bouwstra JA, Gooris GS, Salomons-deVries MA, van der Spek JA, Brasc W, Structure of human stratum corneum as a function of temperature and hydration: A wide-angle X-ray diffraction study, International Journal of Pharmaceutics 1992;84(3):205-216
- Ghyczy M, Nissen H-P, Biltz H, The treatment of acne vulgaris by phosphatidylcholine from soybeans, with a high content of linoleic acid, J Appl Cosmetol 1996;14:137-145
- Lautenschläger H, Das ABC der Fettsäuren, Beauty Forum 2009;12:40-47
- Proksch E, Ungesättigte Fettsäuren. In: Korting HC, Sterry W (Hrsg.), Therapeutische Verfahren in der Dermatologie: Dermatika und Kosmetika, Verlag Blackwell Berlin 2001:183-188
- Lautenschläger H, Spezielle Wirkstoffe und Grundlagen in der Korneotherapie, Kosmetische Medizin 2004;2:72-74
- Lautenschläger H, Angewandte Korneotherapie in der Hautpflege - ein Leitfaden für die Anti-Aging-Behandlung, Ästhetische Dermatologie (mdm) 2007;3:8-16
- Lautenschläger H, Vitamine in der Kosmetik, 2014;10:18-21 und 11;38-40 und 12:34-36
- Lautenschläger H, Radikalfänger - Wirkstoffe im Umbruch, Kosmetische Praxis 2006;2:12-14
- BfR = Bundesinstitut für Risikobewertung
- Lautenschläger H, Periorale Dermatitis - Gründe, Behandlung und Abgrenzung, Kosmetik International 2015;9:44-47
- Lautenschläger H, Weihrauch - Harz mit Heilkraft, medical Beauty Forum 2015;4:12-16
- Lautenschläger H, Cellulite von A-Z, Kosmetische Praxis 2011;1:10-13 und 2011;2:10-12
- Lautenschläger, Flavone und Isoflavone - die Wirkstoff-Generalisten, Kosmetik International 2016;10:62-65
- Lautenschläger H, Kettenreaktion - Hautenzyme und Enzymdefekte, Beauty Forum 2017;1:52-55
- Lautenschläger H, Wasserhaushalt der Haut - Moisturizer & Co., medical Beauty Forum 2014;1:18-20
- Schaefer-Korting M, Korting HC, Kleuser B, Gerber R, Verwendung von Sphingosin, Sphingosin-1-Phosphat, Sphingosin-1-Phosphat-Derivaten und/oder deren Gemischen zur Behandlung von entzündlichen Hautkrankheiten, Dermapharm GmbH, DE 19810999 A1 (13.3.1998)
- Lautenschläger H, Nervensache - erwünschte und unerwünschte Effekte, Kosmetik International 2013;2:40-42
- Lautenschläger H, Bewährter Aufheller - Tranexamsäure wirkt gegen Pigmentflecken und Rötungen, medical Beauty Forum 2015;3:12-14
- Zeisel SH, Choline: an essential nutrient for humans, Nutrition 2000;16:669-671
- Costa KA, Niculescu MD, Craciunescu CN, Fischer LM, Zeisel SH, Choline deficiency increases lymphocyte apoptosis and DNA damage in humans, Am J Clin Nutr. 2006;84;1:88-94
- Lautenschläger H, Vorteile von Produkten ohne Wasser und Hilfsstoffe, Kosmetik International 2017;6:56-58
- Lautenschläger H, Phosphate: Power-Lieferanten für Haut und Haar, Kosmetik International 2017;10:56-58
- Verhoven B, Schlegel RA, Williamson P, Mechanisms of phosphatidylserine exposure, a phagocyte recognition signal, on apoptotic T lymphocytes, Journal of Experimental Medicine 1995;182(5):1597-601
- Lentz BR, Exposure of platelet membrane phosphatidylserine regulates blood coagulation, Prog Lipid Res. 2003;42(5):423-438.
- Mäder K, Klein M, Mauch S, Ramos G, Hofmann U, Meister A, Phosphatidylserine enriched phospholipids as anti-inflammatory agents, Vortrag anlässlich des Symposiums: Phospholipids in Pharmaceutical Research, Heidelberg 18.9.2017
- Birge RB, Boeltz S, Kumar S, Carlson J, Wanderley J, Calianese D, Barcinski M, Brekken RA, Huang X, Hutchins JT, Freimark B, Empig C, Mercer J, Schroit AJ, Schett G, Herrmann M, Phosphatidylserine is a global immunosuppressive signal in efferocytosis, infectious disease, and cancer, Cell Death and Differentiation 2016;23:962-978
- Kim HY, Huang BX, Spector AA, Phosphatidylserine in the brain: metabolism and function, Prog Lipid Res. 2014(Oct);56:1-18
- MMP = Matrix-Metalloproteasen
- Cho S, Kim HH, Lee MJ, Lee S, Park CS, Nam SJ, Han JJ, Kim JW, Chung JH, Phosphatidylserine prevents UV-induced decrease of type I procollagen and increase of MMP-1 in dermal fibroblasts and human skin in vivo, J Lipid Res. 2008(Jun);49(6):1235-45.
- Lautenschläger H, Den Brand löschen - Entzündungshemmende Wirkstoffe, Kosmetik International 2014(2):26-29
- Souci SW, Fachmann E, Kraut H, Food Composition and Nutrition Tables, Medpharm Scientific Publishers Stuttgart (2008)
- Fahr A, Special colloidal structures with phospholipids: Cochleates, 5th International Symposium on "Phospholipids in Pharmaceutical Research", Heidelberg 2017
Dr. Hans Lautenschläger |

