Polyethylene glycols - or abbreviated PEGs - are synthetic raw materials and hence exactly the contrary of what you generally expect in natural cosmetics. Correspondingly, the opinions on benefits and effects also vary. In the end however the physiological features of a substance class are the crucial point in skin care. In this context though facts about specific PEGs do not always apply to the entire substance group. PEGs - endless chains PEGs originate from the inexhaustible model kit of the chemists. The basic module here is ethylene oxide (EO) which is a reactive molecule (C2H4O) that can attach to water, alcohol and carboxylic acids and thus is able to form chains of different length. In combination with water EO forms ethylene glycol which is a popular frost protection agent. Ethylene glycol can continue reacting with EO and hence form diethylene glycol and finally polyethylene glycols (PEGs) of different chain length and consequently also with multifaceted properties.
Diethylene glycol has become well-known to the public due to a series of food and pharmaceuticals scandals (for tolerance data, see reference1)).
Similar to its reactivity with water EO reacts with alcohols. As the reactions are difficult to control various chains of different length develop. The INCI denomination steareth-10 for instance means that 10 EO units on average are linked with the original stearyl alcohol. In other words, steareth-10 may contain varying portions of stearyl alcohol, steareth-1 up to steareth-15 or even higher. The affix "eth" is a general abbreviation for the oxyethylene group (‑O‑CH2‑CH2‑). Anaesthetizing potential PEGs in general are compounds with different components with quite varying properties. A typical example here is laureth-9 which is used as an emulsifier in cosmetics and as a local anaesthetic in dermatology. Laureth-9 (INN: polidocanol) is gained from lauryl alcohol which is a fatty alcohol. Laureth-9 reduces the experience of pain in the skin. Germany's Federal Institute for Risk Assessment (BfR) assesses cosmetics respectively skin care products which contain leave-on substances with anti-pruritic or analgesic features as a potential health hazard for the consumer because the locally anaesthetizing features of the substance will suppress symptoms and alarm signals of the skin like itching and sensation of pain which generally indicate a damaged skin2). Laureth compounds with varying chain lengths are widely used in cosmetics which means that laureth-9 can be contained in any product. It should be added here that varying chain lengths can also occur in the alcoholic part and not only in the EO portion. Among others lauryl alcohol (C12H25OH) frequently also contains myristyl alcohol (C14H29OH), which means that laureth also contains myristeth. The frequently used ceteareth is based on cetearyl alcohol which is a mixture of cetyl alcohol (C16H33OH) and stearyl alcohol (C18H37OH). Subsequently ceteareth is a mixture of ceteth and steareth. Mixtures of different source alcohols also conceal behind the term pareth. C12-C15 pareth-9 for instance has branched synthetic fatty alcohols with 12 to 15 C-atoms like trimethylnonyl alcohol (C12H25OH; isomer with lauryl alcohol), which were converted with 9 EO on average.
In cosmetics PEGs are used as surface active additives. Depending on their composition they are applied as solvents, emulsifiers, tensides, re-fattening agents or moisturizers in skin care as well as cleansing products. Their advantage are their reasonable price and the fact that they can be synthetically produced and exactly adjusted to the features needed in the product. Protection is important Chemically speaking, PEGs are polyethers and just like diethylether which has a similar structure and which is usually labeled as "ether", they are sensitive to oxygen. In the presence of UV light and metal traces they form highly reactive hydroperoxides4-6) and every chemist is well aware of the explosive potential of "ether". That is why it is recommended to store PEG in a dark place and well protected against atmospheric oxygen with antioxidants and chelating agents. The latter mentioned to which the complexing agents EDTA and phytic acid belong to will inactivate the omnipresent metal traces as e.g. iron compounds. This kind of protection is not only important for the raw material itself but also for the finished cosmetic product later on as the hydroperoxides formed in combination with atmospheric oxygen are highly aggressive and reported to be one of the potential causes of the feared Majorca acne7). It is therefore advisable to keep away from direct sun exposure after the application of a PEG containing product as in this case the formation of hydroperoxide is not only influenced by UV light and traces of heavy metals but also accelerated by heat. Based on these facts it seems quite a mystery why PEGs still are used in sun protection products. Fig: Formation of hydroperoxides from PEG
(R1 and R2 represent different remainders of PEG chains) 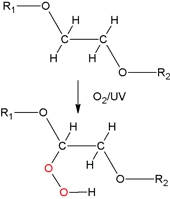
Penetration and permeation
A permeation of PEGs through the intact skin is already minimal with smaller chain lengths, PEGs with extended chain lengths however will not permeate at all. There are different reports regarding the metabolization of PEGs. While a degradation of glycol units to oxalic acid has been stated in some cases most of the authors assume that apart from the cleavage of PEG esters into PEG and fatty acids, PEGs as such are not metabolized and discharged unchanged8). It is not surprising however that there is increased permeation with barrier disorders, dermatoses respectively open wounds and burns.
There is no evidence for a generic penetration supporting effect of PEGs on other substances. An increased permeation9) of ethoxilated oleyl alcohol (oleth-5, oleth-10) can be stated which is already immanent in the features of its basic substances oleyl alcohol (C18H35OH) and oleic acid10) and which also is the reason why the substances are combined with pharmaceuticals.
As expected, there is also an increased penetration with medium sized chain lengths of the underlying alcohol. The maximum is around C12, i.e. in the above mentioned laureth11). Otherwise they are indifferent or there is even a retarded penetration to observe. Compared to anionic and cationic emulsifiers the permeation of ethoxilated alcohols generally is much more pronounced12). At the same time the skin tolerance also is improved. Well tolerated Also the introduction of EO chains in anionic emulsifiers is accompanied with a reduced irritating potential, as for instance can be observed with the transition from sodium lauryl sulfate to sodium laureth sulfate. With lengthening the EO chain also the tolerance will be further improved. At the same time the swellings of the stratum corneum which are a clear indication for the barrier disorder that has been triggered will be reduced13). The skin swelling caused by polysorbate-20 is only the fiftieth fraction of that of sodium lauryl sulfate which has tenside features and which is routinely used as a standard irritant in skin tolerance measurements.
A significant fact in this context also is the critical micelle concentration (CMC) - the lower the better the tolerance. The background here is that the free molecules carry the aggressive potential and less their micelles. The CMC of sodium lauryl sulfate is 7.2 mmol/l and of sodium laureth-3 sulfate 0.70 mmol/l. This means that the skin swelling caused by sodium laureth-3 sulfate is only a quarter of that caused by sodium lauryl sulfate14). Biological degradation The fact that PEGs are resistant to chemical and microbiological degradation is an advantage for the stability and storage of cosmetic and dermatological products. Used as emulsifiers in creams their disadvantage compared to physiological and surface active compounds like monoglycerides, diglycerides, carboxylic acid/sodium and potassium salts is that their emulsifying activity remains unchanged in the skin. In other words: similar to other synthetic non-degradable emulsifiers they can cause washout effects during skin cleansing, even of natural skin lipids. This specific feature can lead to long term intolerances in combination with atopic skin as its recovering ability is comparably low anyhow15).
In connection with PEG the following substances are of particular importance: Dioxane: 1,4-Dioxane is formed by reaction of 2 molecules of ethylene glycol. In the eighties this substance was found as a by-product of PEGs and hence became a major issue in press reports. According to the Cosmetic Decree (KVO) 1,4-dioxane is banned from cosmetics. Ethylene oxide: in the Cosmetic Decree also listed as a banned substance. Heavy metals: as the case may be, PEGs may cause an increased absorption of heavy metals. Heavy metals support the autoxidation. Specifically make-ups that are applied over a PEG-containing skin care should be free of ferreous pigments. Iron ions are physiologically harmless but support the formation of hydroperoxides. In organic chemistry metal compounds of PEGs and their related cyclic polyethers (crown ether) are used as reaction accelerators (see chemistry textbooks).
PEG and related compounds
As mentioned above, the substance class of PEGs and PEG relatives includes a large variety of individual substances which, depending on their structure, practically cover all the relevant HLB range16) with regard to their surface activity. In the following the most important representatives are listed in alphabetical order. APG: like PEGs alkylpolyglycosides belong to the polyethers with the glycol units being replaced by glucose. Specifically in shampoos APGs distinguish themselves by their excellent feeling on the skin. They can be used in microemulsions. Carbowax: other term for PEGs and methylated PEGs (MPEG) which are mainly used in ointments and suppository bases and as additives in pills. Macrogols (INN): PEGs that are used as laxatives in medicine. They are neither absorbed nor degraded by the intestinal flora and hence discharged unchanged. To characterize them their molecular weight is added behind the technical term as e.g. macrogol 400 with a molecular weight between 380 and 420. What macrogols are in the field of medicine that are PEGs in cosmetics. Macrogol 400 is identical with PEG-9, i.e. a polyethylene glycol consisting of 9 EO units. Macrogol 4000 is a laxative. The mixture of 60 percent by weight of macrogol 400 and 40 percent by weight of macrogol 4000 is a typical base for ointment formulations in the pharmacy. PEG: characteristics as described above. Further details: Lit.17). PEG beeswax: Gelling agent in oleogels. The degree of ethoxilation is indicated with a hyphen behind the PEG and can amount from 3 to 20. PEG fatty alcohol ether: characteristics as described above. PEG lanolin: is gained by ethoxilation of lanolin. PEG-75 lanolin e.g. is used as re-fattening agent in cleansing products like shampoos and shower creams. Besides PEG-75 there is a multitude of additional degrees of ethoxilation. PEG esters: are gained by ethoxilation of carboxylic acids. In this process monoesters as well as diesters may form. PEG-150 distearate e.g. is a PEG-150 which is esterified with stearic acid at both ends of the chain and used as thickening agent in shampoos. PEG glycerides: PEG-6 caprylic/capric glycerides is a monoglyceride gained from a mixture of caprylic and capric acids ethoxilated with 6 EOs on average. PEG-15 glyceryl oleate is an ethoxilated monoglyceride of the oleic acid, only to mention two examples out of a multitude of different forms. PEG (Hydrogenated) Castor Oil: hydrogenated castor oil can be linked to a few or many EOs. Similar to other PEGs the degree of ethoxilation is indicated with a hyphen behind the PEG. This substance class is frequently used in W/O emulsions. Poloxamer: ethoxilated PPG ethers (polypropylene glycol ethers) also called EO/PO block polymers. The EO or PO content cannot be derived from their INCI denomination. Poloxamer-212 e.g. is a PPG-35 which is linked with 8 EOs on both sides of the molecule. Polyglycerins: polyethers of glycerin, with increased hydrophilic property due to a free hydroxyl group per glycerin unit. By reaction with acids polyglyceryl esters will generate as e.g. polyglyceryl-3 diisostearate and polyglyceryl-3 oleate. Polyglycerin derivatives are the main component of niosomes which are the synthetic alternative to liposomes (main component phosphatidylcholine [PC]). With appropriate formulations of PEGs, polysorbates and sorbitan esters, cell-like vesicles can be generated. In contrast to the PC of liposomes the components of niosomes are either not or only marginally degraded in the skin. Polysorbates: the above mentioned polysorbate-20 is a sorbitan whose 4 OH groups are ethoxilated. The total number of linked EOs is 20. These can again be esterified at their ends with the result that surface active products are generated which became known as Tween. PPGs: analogue to PEGs, polypropylene glycols are gained from propylene oxide (PO). Their denomination also corresponds to the PEG-terms: PPG-9 contains 9 POs for instance. PPG-3 myristyl ether is produced from myristyl alcohol and 3 PO units. By nature, PPGs are more hydrophobic than PEGs and this is the reason why they are partly used as replacement for mineral and vegetable oils. Sodium Laureth Sulfate: contains 1 to 4 EO units and is mainly used in shampoos and cleansing products. Ether sulfates combine the properties of anionic and non-ionic tensides. Sodium Laureth Phosphate: The degree of ethoxilation is shown after the hyphen as e.g. sodium laureth-4 phosphate which is applied in cleansing products and as emulsifier in creams.
In addition to that there are various other combinations as e.g. ethoxilated soy sterols, (PEG soy sterol), PEG-PPG-carboxylic acid esters, ethoxilated amides and amines. As the characteristics depend on the degree of ethoxilation it is impossible at this point to give a general answer regarding their use. The more dominant the polar ether groups or in case of polyglycerins the additional hydroxyl groups the more hydroscopic or water-attracting the PEGs become. Higher molecular PEGs form moisture-retaining films on the skin surface with minimal influence on the skin barrier layers as they do not penetrate into the skin. It should be kept in mind however that an appropriate protection against heavy metal traces and UV radiation is needed. Glossary CMC: abbreviation for Critical Micelle Concentration. The CMC indicates the concentration of free molecules of a surfactant. If the concentration is exceeded the free molecules aggregate to so called micelles, frequently with spherical structure. The lower the CMC the better the tolerance of the substance. Emulsifiers: stabilize fats and oils in drop-like form in creams and lotions. HLB: abbreviation for Hydrophilic-Lipophilic-Balance. Every surfactant has been allocated a certain HLB value on the HLB chart which results from its hydrophilic and its lipophilic portion. Consequently, the average values for foam-inhibiting substances are 1.5 to 3, for W/O emulsifiers 3 to 8, for surfactants 7 to 9, for O/W emulsifiers 8 to 18, for tensides 13 to 15 and for solvents for aqueous systems 12 to 18. INN: abbreviation for International Nonproprietary Name. Every pharmaceutical agent has an unmistakable, internationally valid denomination. Isomer: chemical compounds which have the same molecular formula (same type and number of atoms) however a different structural formula, i.e. the atoms are differently linked. Example: C12 pareth-9 and laureth-9, this is the reason why they also have different INCI denominations. Leave-on products: cosmetics which stay on the skin after their application as e.g. skin care creams. Pharmaceutical formulations: preparation of pharmaceuticals in the pharmacy Microemulsions: emulsions in which interfaces like drop-like structures can no longer be determined by microscopic magnification Penetration: infiltration of substances into the stratum corneum and other surface near skin layers Permeation: passage of substances through the skin into the body. Rinse-off products: cosmetics which are completely rinsed off after their application Re-fattening agents: surface active components in cleansing products which stay on the skin after the cleansing and then form a fat-like film Nonionics: abbreviation for non-ionic emulsifiers and tensides. Besides PEGs and their related PPGs, also polyglycerins, APGs and polysorbates belong to this group.
References (not contained in the original publication)
Abstract Toxikologische Bewertung Nr. 11, Diethylenglykol 12/95, Berufsgenossenschaft der chemischen Industrie, Heidelberg Polidocanol in kosmetischen Mitteln, Stellungnahme des Bundesinstituts für Risikobewertung (BfR) vom 15.Oktober 2003 AAPS PharmSciTech 7 (3), E47-E53 (2006) Hager's Handbuch der pharmazeutischen Praxis von F. von Bruchhausen et al, Springer Verlag, Heidelberg 1994, S. 290 (Polysorbat-20) Fiedlers Encyclopedia of Excipients of Pharmaceuticals, Cosmetics and Related Areas, 1096-1098, Editio Cantor Verlag, Aulendorf 2007, 1148-1153 R. Hamburger, E. Azaz und M. Donbrow, Pharm Acta Helv. 50, 10-17 (1975) Tronnier H, U. Heinrich, Die Mallorca-Akne: Follikulär-papulöse Lichtdermatose als eigenes Krankheitsbild - aktueller Wissensstand, SÖFW-Journal 2000 (4) 54-55 (zitiert in GD Gesellschaft für Dermopharmazie e. V.: GD-Leitlinie "Dermokosmetischer Sonnenschutz" in der Fassung vom 23.11.2007). C. Fruijtier-Pölloth, Safety assessment on polyethylene glycols (PEGs) and their derivatives as used in cosmetic products, Toxicology 214 (2005) 1-38 S.-C. Shin, C. W. Cho und I. J. Oh, Effects of non-ionic surfactants as permeation enhancers towards piroxicam from the poloxamer gel through rat skins, Int. J. Pharm 222 (2), 199-203 (2001) Surfactants in Cosmetics, M.M. Rieger und L. D. Rhein, Verlag Marcel Dekker, Inc. 1997, S.514 Surfactants in Cosmetics, M.M. Rieger und L. D. Rhein, Verlag Marcel Dekker, Inc. 1997, S.513 Surfactants in Cosmetics, M.M. Rieger und L. D. Rhein, Verlag Marcel Dekker, Inc. 1997, S.564 Surfactants in Cosmetics, M.M. Rieger und L. D. Rhein, Verlag Marcel Dekker, Inc. 1997, S.400 Surfactants in Cosmetics, M.M. Rieger und L. D. Rhein, Verlag Marcel Dekker, Inc. 1997, S.403 H. Lautenschläger, Pflege bei Neurodermitis, Kosmetische Praxis 2005 (1), 9-11 Fiedlers Encyclopedia of Excipients of Pharmaceuticals, Cosmetics and Related Areas, 1096-1098, Editio Cantor Verlag, Aulendorf 2007, 112-119 Fiedlers Encyclopedia of Excipients of Pharmaceuticals, Cosmetics and Related Areas, 1096-1098, Editio Cantor Verlag, Aulendorf 2007, 1148-1153.
Dr. Hans Lautenschläger |

